BioTeSys – Nutritional CRO & Test laboratory
BioTeSys has been securing and testing your products as well as supporting you on questions related to bioactive ingredients for more than 25 years.
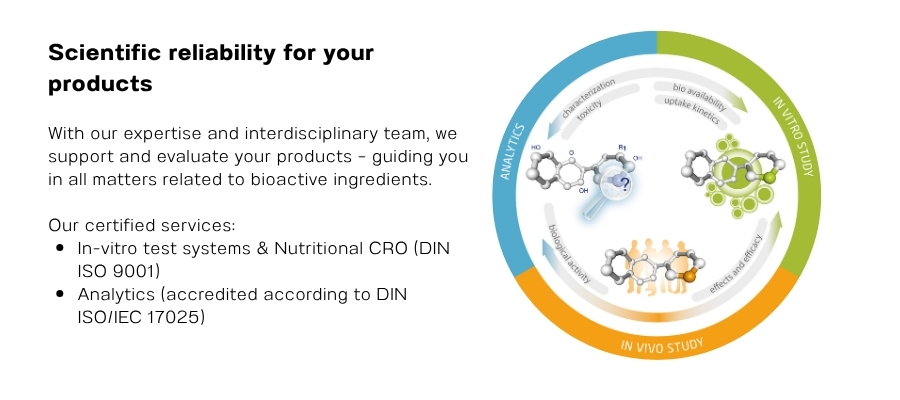
Your specialist for scientific proof of the effects, efficacy, and safety of food, cosmetics, and medical devices
We specialize in the scientific validation of food, cosmetics, and medical devices, combining analytical and cell-based methods with complex clinical studies to rigorously assess product properties – from bioavailability, effect, and efficacy to safety and regulatory compliance. Leveraging our interdisciplinary expertise, we design tailored study protocols that optimize both time and budget for our clients. This ensures a robust basis for product development, approval, and successful market launch.
Strategic Partnership: Biotesys and Clinic'n'Cell Unite Expertise in Nutraceutical Research
We are excited to announce our collaboration with Clinic'n'Cell. Our competencies in clinical studies for nutraceuticals perfectly complement Clinic'n'Cell's innovative ex vivo research approach. Our shared goal: To elevate the connection between human metabolism and cellular biology to an entirely new level. This partnership marks a significant step in nutraceutical research and enables innovative approaches in medical science. We look forward to this inspiring collaboration!
moreBioTeSys commits to SBTi and invests in global emissions reductionst
Decarbonization always begins with reducing emissions. BioTeSys is facing up to its responsibility and striving to make a positive contribution by following the Science Based Targets Initiative (SBTi) and investing in projects that help to reduce emissions worldwide.
moreCollaboration with daacro: Together into an innovative future
Cooperation with daacro: BioTeSys is pleased to announce a collaboration with the daacro research institute. This collaboration opens up new opportunities to join forces, create synergies and realize groundbreaking projects together. With daacro's know-how in the field of psychotropic substances and BioTeSys' expertise in analytics and in vivo studies, we are laying the foundation for innovative developments and progress in our areas of expertise. The goal of the collaboration is to better meet the needs of our customers through an intensive exchange of knowledge and resources. Together, we look to the future with confidence and look forward to achieving important advances for science through this partnership. Link to daacro
BioTeSys joins CERTANIA group
BioTeSys, a premier Contract Research Organization (CRO) specializing in the evaluation and testing of bioactive ingredients for nutraceuticals, cosmetics, and medical devices, proudly announces its integration into CERTANIA, a dynamic group focused on Testing, Inspection, and Certification (TIC) services. This strategic integration marks a significant milestone for BioTeSys, enhancing its ability to deliver comprehensive research and testing solutions across diverse sectors while acting as a "one-stop-shop" for its clients.
learn moreAllègement fiscal pour les travaux de R&D des partenaires français de BioTeSys
L’entreprise BioTeSys GmbH est agréée au titre du crédit d’impôt recherche, CIR, par le Ministère de l’Enseignement Supérieur, de la Recherche et de l’Innovation, en qualité d’organisme prestataire de services en R&D. Nos clients français bénéficient d’un allègement fiscal allant jusqu’à 30% des dépenses engagées en recherche et développement dans le cadre de leur projet.
learn moreCANNABIS-NET: a German-Canadian research and development cooperation
BioTeSys is involved in the "Medical phytocannabinoid-rich (PCR) Cannabis" project. Goal of this project, initiated by an international research network of research facilities and companies in Germany an Canada, is to lead new developments and innovations with the aim to improve the field of medical application of cannabis and PCR cannabis.
learn more